Drug Patent Cliff is a Catalyst for Biotech
March 19, 2026
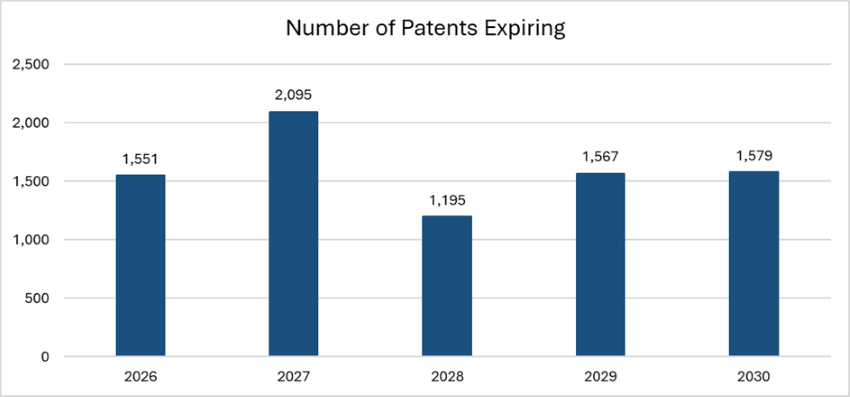
As highlighted in today’s Chart of the Week, approximately 8,000 drug patents are scheduled to expire by the end of 2030.1 As a result, the U.S. pharmaceutical market is expected to lose $230 to $300 billion in revenue between 2025 and 2030.2,3 Once exclusivity lapses, generic manufacturers can enter the market, and for small-molecule drugs in particular, price competition tends to be swift. In the U.S., prices can drop by one-third in the first year after expiration and by more than 80% within eight years.4
Patent expirations, therefore, create a powerful incentive for pharmaceutical companies to replenish pipelines, either through internal research and development (R&D) or external acquisition. Internal development remains expensive, with an average drug development cost of approximately $2.2 billion per asset.5 Given these economics and the long lead times associated with organic R&D, mergers and acquisitions (M&A) and structured partnerships have become core strategic tools rather than one-off options. While the number of biopharma M&A deals in 2025 was broadly similar to 2024, total deal value rose sharply—from $81 billion to nearly $200 billion—signaling a shift toward larger, more strategic acquisitions.6 Recent examples include Merck’s acquisition of Cidara Therapeutics for $9.2 billion and Novartis’ $12 billion purchase of Avidity Biosciences.7
The therapeutic areas attracting significant amounts of capital—oncology, immunology and metabolic disease—reflect both scientific momentum and significant commercial whitespace. Each combines rapid advances in biology and modality innovation with large, underserved patient populations and multi-billion-dollar revenue pools at risk from loss of exclusivity. In oncology, continued breakthroughs in modalities such as antibody-drug conjugates and cell therapies have sustained high deal activity. In immunology, a growing understanding of autoimmune and inflammatory pathways is enabling more targeted therapies that can displace aging biologics. In metabolic disease, the success of GLP-1s has unlocked broader investment across obesity and cardiometabolic indications, creating new opportunities beyond first-generation assets.
Partnership activity via multi-asset licensing arrangements has also been accelerating alongside outright acquisitions, reflecting a growing preference for shared-risk structures. Large pharmaceutical companies are also increasingly pursuing smaller, earlier-stage transactions alongside traditional late-stage or de-risked assets, seeking access to novel platforms, differentiated biology and optionality rather than single-asset replacement strategies. This approach allows acquirers to spread risk while accelerating exposure to innovation that would be difficult to replicate internally.
Key Takeaway
With approximately 8,000 patents expiring in the next five years, the pharmaceutical industry faces a period of transition that will require active pipeline management and disciplined capital allocation. As a result, the patent cliff is acting as a catalyst for tighter integration between large pharma and the biotech ecosystem. Capital is moving earlier in the drug-development lifecycle, providing liquidity and clearer exit pathways for investors while recycling proceeds back into discovery-stage innovation. This virtuous cycle is compressing the historical timelines between discovery, validation and commercialization, shifting value creation away from aging blockbusters and toward differentiated biology and scalable platforms.
Sources:
1U.S. Food & Drug Administration – Orange Book Data Files; 2/14/26
2Drug Discovery News – Blockbuster drugs face a massive patent cliff in 2026; 2/24/26
3DeepCeutix – $300 Billion in Pharma Revenue Loses Patent Protection by 2030; 2/2/26
4JAMA Health Forum – Drug Prices After Patent Expirations in High-Income Countries and Implications for Cost-Effectiveness Analyses; 8/16/24
5Deloitte – Measuring the return from pharmaceutical innovation; March 2025
6,7Nature Reviews – Biopharma dealmaking in 2025; 1/21/26
This material is for informational use only. The views expressed are those of the author, and do not necessarily reflect the views of Penn Mutual Asset Management. This material is not intended to be relied upon as a forecast, research or investment advice, and it is not a recommendation, offer or solicitation to buy or sell any securities or to adopt any investment strategy.
Opinions and statements of financial market trends that are based on current market conditions constitute judgment of the author and are subject to change without notice. The information and opinions contained in this material are derived from sources deemed to be reliable but should not be assumed to be accurate or complete. Statements that reflect projections or expectations of future financial or economic performance of the markets may be considered forward-looking statements. Actual results may differ significantly. Any forecasts contained in this material are based on various estimates and assumptions, and there can be no assurance that such estimates or assumptions will prove accurate.
Investing involves risk, including possible loss of principal. Past performance is no guarantee of future results. All information referenced in preparation of this material has been obtained from sources believed to be reliable, but accuracy and completeness are not guaranteed. There is no representation or warranty as to the accuracy of the information and Penn Mutual Asset Management shall have no liability for decisions based upon such information.
High-Yield bonds are subject to greater fluctuations in value and risk of loss of income and principal. Investing in higher yielding, lower rated corporate bonds have a greater risk of price fluctuations and loss of principal and income than U.S. Treasury bonds and bills. Government securities offer a higher degree of safety and are guaranteed as to the timely payment of principal and interest if held to maturity.
All trademarks are the property of their respective owners. This material may not be reproduced in whole or in part in any form, or referred to in any other publication, without express written permission.

